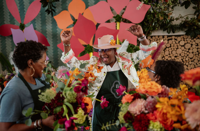Atlantic Station and the Car Park, a parking management company, have announced a long-term partnership effective in May.
The Riverwood boys’ lacrosse team appears to be peaking at the right time as it enters the Class 5A-6A state playoffs.
Sandy Springs has approved $146,000 in grants for nonprofit arts and recreation programs targeting the city’s underserved and low-income communities.
A new museum is being planned for Downtown Atlanta, where it will be America’s only museum of the future.
Trending
Earth Day fun facts for kids and adults.
The Taste of Alpharetta, Metro Atlanta's longest-running food festival and a highlight event of the Southeast Tourism Society, will draw culinary enthusiasts to downtown Alpharetta on May 9. Featuring free admission, the festival offers attendees a chance to savor dishes from over 60 local r…
Buckhead Village's third annual Bodacious Blooms Flower Festival is slated to make a lively return May 2 through May 4.
Trending Now
-
Grady to Build Much-Needed Emergency Facility in Union City
-
Riverwood Boys’ Lacrosse Overcome Early Struggles to Make State Playoffs
-
New Sandy Springs Wrestling Center Brings a New Era of Inclusivity to Northside
-
2 Men Injured, 1 Stabbed in Robbery at Buckhead Apartment Complex
-
Postmaster General Dejoy Put in Hot Seat as Ossoff Pushes for Answers
3540 Woodhaven Road has 11 bedrooms, 13 full and 7 half bathrooms and is on the market for $35,000,000 Click here for all the photos.ON THE MARKET: A Tuxedo Park Masterpiece in the Heart of Buckhead
On The Market:

News
Painted Pickle, a new dining, gaming and pickleball concept, has opened at Armour Yards in between Buckhead and Midtown.
Ashlyn Fuller will never forget the day she fell in love with wrestling.
U.S. Senator Jon Ossoff slammed Postmaster General Louis Dejoy in a U.S. Senate Homeland Sec…
Marsy’s Law for Georgia has honored and recognized Fulton County Court Clerk Ché Alexander f…
The College Park Skyhawks allowed fourth and fifth-grade West Clayton Elementary School stud…
The city of Hapeville celebrated the 100-year anniversary of the Hapeville Fire Department a…
Mayor Vince Williams and the City Council came together to bring Union City an energetic Sta…